The CDC’s new quarantine and isolation rules were what pushed me to publish today. But I'll also offer you big treatment and vaccine news, discouraging Omicron updates, graphic vaccination lessons, and more.
Treatment news
 |
| Convalescent plasma rides again |
Paxlovid: The most exciting update is the FDA authorizing Pfizer’s pill for emergency use in high-risk COVID-19 outpatients, and the EMA allowing European Union members to do so if they wish. Paxlovid has been shown to cut the risk of hospitalization and death in this group by nearly 90% if a 5-day course is started within 5 days. Plus – more big news – a trial in standard-risk patients seems to be showing 70% fewer bad outcomes. This could transform the shape of the pandemic, but I have several caveats. First, as yet we don’t know for sure it works against Omicron. Second, though it doesn’t have the serious risks associated with molnupiravir, not everyone can take it – not patients with liver or kidney disease, or on medications ranging from the antiinflammatory piroxicam to certain antiarrythmics, sleeping pills, anticonvulsants, even Saint John’s Wort. Many of those drugs can be safely paused for 5 days, though, to allow for Paxlovid trreatment.
Finally, to have a real impact on the pandemic a drug needs to be readily available worldwide. Given major supply shortages the pill may arrive too late to flatten the curve of the Omicron surge – Italy won’t have any before February or even March. And Pfizer must keep its promise to lower prices from the current $529 per course if Paxlovid is to be within reach of middle- and low-income countries. Distribution issues can be expected even in some wealthy countries, notably the United States, whose chaotic healthcare non-system makes access to testing and physicians difficult for many people. This may change when Biden’s half-billion free home test kits get – eventually – into the hands of American families. Paxlovid is arguably the best single reason for home testing. (Assuming those tests can detect Omicron, which is not guaranteed.)
Molnupiravir: Merck’s pill received an Emergency Use Authorization from the FDA the day after Pfizer’s, but I doubt its executives are happy. The FDA has demanded considerable further post-marketing research, and wisely specifiesthat this drug should be used only if “alternative COVID-19 treatment options authorized by FDA are not accessible or clinically appropriate.” Paxlovid and anti-Omicron monoclonal antibodies should soon arrive, so molnupiravir will likely be used very little…
…if at all. The full Phase 3 trial results for molnupiravir have now been published, and the difference between the mediocre interim results and the frankly poor final results stands out like a sore thumb. The authors tried hard to explain it away as due to “imbalances between the analysis samples, shifts in the epidemiology of the Covid-19 pandemic, and regional variation among the enrolled participants.” But the real reason is inefficacy against the Delta variant: buried in a supplementary appendix is the crucial information that the drug was only 24% effective against Delta but 60% effective against other strains. Oddly, the New England Journal of Medicine editorialist didn’t pick up on this. Between efficacy that’s poor against Delta and unknown against Omicron this drug is next to useless. I’ve even submitted a letter to the editor, what the hell.
Even worse news for Merck from France: it’s cancelled its order for 50,000 pills. I wish other countries would do the same, but Italy and Germany have instead confirmed theirs, and a first shipment has already arrived on Italian soil. We’ll see what gets done with it.
Monoclonal antibodies: Both the Lilly and Regeneron products have been shown by a Mayo Clinic study to be effective against Delta not just in unvaccinated patients, but also in high-risk vaccinated people with breakthrough infections, lowering the hospitalization rate by 77%. This was a retrospective study, though, and we can’t be sure without placebo-controlled clinical trials. Sotrovimab, the antibody expected to be most effective against Omicron, wasn’t examined. The US government is now frantically stockpiling sotrovimab, which has been scarse there and practically nonexistent in Europe.
Before Christmas the US Department of Health and Human Services stopped distributing monoclonal antibodies from Lilly and Regeneron on grounds of their probable inefficacy against Omicron. But this was based on the umpteenth screwup from the CDC, which had claimed that 73% of new cases in the US were due to Omicron. The next week it turned out the real figure is not 73% but……23%! This revision, or rather admission of incompetence, means Delta is still by far the dominant strain in the US and Lilly and Regeneron are still invaluable. Hopefully that distribution will restart pronto.
Remdesivir: A study I’ve mentioned showing remdesivir can help outpatients has now been published: 87% efficacy at preventing hospitalization or death at 28 days, among high-risk unvaccinated patients treated within a week. This would be grand, if not that 1) we don’t know whether it works in vaccinated people, and 2) patients have to drag themselves in 3 days in a row for lengthy intravenous infusions. Since monoclonal antibodies can be given in a single dose, and the Paxlovid pill will be even more convenient, outpatient remdesivir will probably be used only in people with contradictions to Paxlovid and no access to monoclonals.
Convalescent plasma: This treatment was discarded months ago but now seems to have risen phoenix-like from its ashes, with a large American study finding a dose of antibody-loaded plasma cut hospitalization rates by 54%, in the Delta era, when given early to unselected COVID-19 patients. The FDA has already acted on these results.
Vaccine news
 |
| Donald Trump gets booed for admitting he had a vaccine booster |
Myocarditis, again: New Zealand is investigating the cases of a 13-year-old boy and a 26-year-old man who died from myocarditis following the Pfizer vaccine. I know of only 2 other such deaths, but the risk of myocarditis is high enough that I continue to advise prudence regarding boosters to males under 30, and in favor of delaying their second doses. In a new study from Denmark Moderna caused 3 times as much myocarditis as Pfizer, so it’s clearly to be avoided in that age group.
Vaccine politics: “Trump was appearing on Bill O'Reilly's ‘History Tour’ at the American Airlines Center in Dallas when the host asked if he was boosted. ‘Yes,’ Trump replied.” This confession was met with boos. If Anthony Fauci was “stunned” that only means he’s out of touch.
Johnson & Johnson: Preliminary evidence from an observational study in South Africa suggests that a second shot protects well against hospitalization due to the Omicron variant. But confirmation from countries with older populations, fewer people exposed to the coronavirus, and higher vaccination rates will be necessary before we start recommending a J&J booster over a Pfizer one.
Corbavax: This new entry, inexpensive and easy to produce, was developed by researchers at the Texas Children’s Hospital and is already approved in India. But there’s as yet minimal proof of its efficacy.
Novavax: Double good news for this company’s vaccine, but important caveats. First, it’s been approved by the European Medicines Agency, becoming the 5th one available in the EU. Caveat: its Phase 3 studies were all completed before Delta and way before Omicron, and it was ineffective against Beta, so we don’t know how well it will perform in the current phase of the pandemic. Some think the conventional technology of Novavax – similar to our normal hepatitis vaccine – will convince those who think “experimental” vaccines will mess with their DNA and spread spike protein around their body to get vaccinated. I have my doubts. Conspiracy theories mutate even more rapidly than coronaviruses.
Also: twin booster trials are now underway. One arm will give a third dose to volunteers in the original Phase 3 trials, while the other will use it as a heterologous booster to other vaccines. Heterologous boosting could be its main use in Italy. Caveat: I hope the booster works better in real life than it did in the lab, when it looked particularly mediocre in people who’d been vaccinated with AstraZeneca or Pfizer.
Moderna meets Omicron: A small company-funded laboratory study has found that two doses of Moderna, like Pfizer, do little against the Omicron variant, but that a third dose gives a big boost to neutralizing antibodies. The researchers conclude that there’s no need to make variant-specific versions of the vaccine. If Moderna throws in the towel, I hope Pfizer doesn’t. Given that neutralizing antibodies don’t necessarily translate into real-life efficacy, that Pfizer’s effectiveness against Omicron seems much less than it was against Delta, and that we have close to zero real-life data for Moderna, am I allowed to say this conclusion pisses me off?
…We do now have two morsels of Moderna data: in a superduperspreader event in Norway, people who had been vaccinated with Moderna were less likely to get sick than those vaccinated with Pfizer, leading the Norwegian Institute of Public Health director to say that, like me, she’d take a Moderna booster after 2 doses of Pfizer. An English government report also suggests Moderna boosters are a bit better than Pfizer in preventing Omicron. But there’s bad news from that same report: boosters’ enhanced protection against Omicron wore off fast, dropping at 10 weeks by 15%-25%.
Pfizer meets Omicron: A study from South Africa suggests that double-dose Pfizer vaccine lowers the risk of hospitalization by 70%, much better than their 33% or so effect on COVID-19 overall. As usual, effectiveness may be different in Western countries.
The cavalry to the rescue: The United States Army has announced to considerable hype that they’ve developed a vaccine good against all SARS-CoV-2 variants including Omicron. Universal vaccines are one of my favored pipedreams but – hold on – human trials of this candidate vaccine are only at Phase 1. And even those results, about short-term safety and immune system stimulation, haven’t been released yet. Their research in monkeys has mostly looked at immune responses, and the one study of protection against disease was in hamsters. Furthermore, the hamsters were given only Alpha and Beta variants, and in monkeys the immune response was particularly weak against Delta. If this vaccine ever hits the market, it will be many months from now.
Infectiousness: A German study has found that people exposed to a fully vaccinated infected individual were about one-third as likely to become infected themselves than if they were exposed to someone who was not vaccinated. While not surprising, this is reassuring.
It wanes, it wanes not: We know that protection against severe COVID-19 stays high for many months after vaccination even as protection against mild or asymptomatic infection falls away. One small but careful study helps to understand why: memory B cells and memory T cells persist and even increase over 6 months, while ordinary anti-spike antibodies tank.
Booster nightmares: In Israel they’ve found that antibodies start falling just a few months after the booster, so they were all set to start giving a 4th dose of Pfizer to healthcare workers and elderly people who had their 3rd doses back in July and August. But now they’re doing a clinical trial instead, definitely a better idea.
Staying home
 |
| The Oslo Christmas party restaurant |
Self-isolation: Here’s its death by 1000 cuts. Infected individuals have always been advised to stay home in complete isolation for 10 days. Before Christmas I was scandalized by new English guidelines cutting self-isolation from 10 days to 7, following two negative antigen swabs. The English authorities themselves didn’t seem fully convinced, if they “strongly advised” those leaving self-isolation early to “limit contact with others in crowded or poorly ventilated spaces, work from home and minimise contact with those most vulnerable to COVID-19.”
Next the Centers for Disease Control approved shortening the self-isolation period to 7 days for healthcare workers. I thought that was a particularly bad idea, since we know people remain infectious for up to 10 days, and hospital patients are the last people we should be exposing to SARS-CoV-2.
Now the CDC has gone one step further in the wrong direction, saying that all swab-positive people whose “symptoms are resolving after 5 days” can be out and around after just 5 days of isolation – merely wearing masks around others for another 5 days. The CDC isn’t even demanding a negative antigen test first, though they do suggest testing “If possible.” And even a negative antigen test is no guarantee a patient is not infectious, especially with Omicron. Good lord.
Of course I understand the motivation for these policy changes. With Omicron on the rampage, previous regulations would have vast numbers of people at home quarantining or self-isolating, risking major personnel shortages in essential workplaces. Lack of staff already has New York City subways running late, half the shows on Broadway shuttered, thousands of flights cancelled, and American hospitals preparing to call retired workers back into service. The National Hockey League season has evaporated and the Miss World competition was postponed when 23 of the 97 contestants came up positive. Omicron is so contagious that 81 out of 117 Christmas party guests at one Oslo restaurant – all swab-negative and almost all double-vaccinated – came down with COVID-19, mostly Omicron, as did 70 unfortunates who happened to be sitting at nearby tables.
I have been able to find zero evidence that Omicron patients are contagious for a briefer period of time. In fact, I suspect the opposite may be the case, that the massive load of virus in the nose could lengthen the period of viral shedding. And numerous reported clusters confirm my clinical impression: Omicron is probably an exception to the rule that vaccinated breakthrough cases are less likely to infect their close contacts.
The justification given by Walensky and those toeing her party line – unfortunately Anthony Fauci is among them – is that people with COVID-19 are most infectious early on: “the majority of transmission occurs early in the course of illness, generally in the one to two days prior to onset of symptoms and the two to three days after.” This is true, but “the majority” is by no means all. Sick patients are usually most infectious within a week of becoming ill, but many remain contagious for 9-12 days (somewat fewer for asymptomatic infections).
I find the new CDC advice horrifying, coming while the pandemic is spinning out of control. As do…
- A professor of epidemiology: “I don't know a lot of people that feel super comfortable with this decision.”
- A university health officer who called his column, “The CDC has new COVID guidelines. This is what it got wrong.”
- A top infectious disease specialist: “It feels to me the wrong time to relax any guidance for the public.”
- A flight attendant union president: “No worker should be forced to come to work when they’re still sick.”
- A professor of internal medicine: “you are in effect encouraging people to engage in activities that could increase transmission.”
- A professor of immunology and infectious diseases: “this feels honestly more about economics than about the science.”
- Michael Mina, Harvard epidemiologist turned rapid-testing apostle: “CDC’s new guidance to drop isolation of positives to 5 days without a negative test is reckless. Some ppl stay infectious 3 days, some 12. I absolutely don’t want to sit next to someone who turned Pos 5 days ago and hasnt tested Neg. Test Neg to leave isolation early is just smart.”
The one exception I’d make to the 10-day rule would be to allow swab-positive hospital personnel to work on COVID-19 wards – even without any self-isolation – if they feel up to it.
Quarantine: The CDC has long advised close contacts of infected individuals to quarantine (for 14 days) only if they’re unvaccinated. The latest update reduces that period to 5 days for the unvaccinated and the vaccinated but unboosted, letting only those who’ve had a booster to forego quarantine altogether. All of them should “Test on day 5, if possible.” Again, good lord – “if possible”? Even worse, everyone who’s unvaccinated or unboosted is told, “If you can’t quarantine you must wear a mask for 10 days.” Huh???? Who exactly falls in that category????
Note that 14 days covers the conventional incubation period for COVID-19, while 5 or even 10 days does not. But the quarantine change, as opposed to the self-isolation change, does have a kernal of validity. With early COVID-19 strains, half of patients became ill more than 5 days after their exposure. With Delta the halfway point fell to about 4 days. The average incubation period with Omicron is even shorter, about 3 days, judging from clusters in Nebraskaand Oslo. That means it is not unreasonable to shorten the period of obligatory quarantine. But not to 5 days! If with previous strains the time between exposure and illness was considered to range up to 14 days (though it was occasionally even longer), about triple the average incubation period, until we get better data we can estimate Omicron’s maximum incubation period at 10 days. If quarantine must be truncated to 5 days it is vital that people test before they emerge from home, and that they wear a well-fitting KN95 mask scrupulously until at least 14 days have passed. Remember: 1) antigen tests often don’t pick up Omicron; 2) Delta, with its longer incubation period, is still the dominant variant in countries including both the United States and Italy. (Italy has softened quarantine regulations too, but only slightly.)
As the situation deteriorates – daily cases in the US and Italy are now triple what they were 10 days ago – what we need is not masses of infected individuals loosed on our streets, but mask mandates, vaccine mandates, distancing, Green Passes, telework, and strict limits on gatherings. What’s coming instead, at least in Republican-leaning US states, is the exact opposite.
My bottom line, and I’ve said it before: Rochelle Walensky is an intelligent and well-meaning person, but she’s in over her head. She should not be running the CDC. These new policies and the recent snafu over percentages of Omicron cases are just the latest proof.
I confess my personal paranoia is starting to match a tweet from Dr. Tatiana Prowell: “The American public needs a clear message. It does not appear to be coming, so here's my best attempt. If you are able to, you should: STAY HOME, STARTING NOW.”
Bad Omicron news
 |
| A positive antigen test |
The latest data from South Africa say that people infected with Omicron are 80% less likely to need hospitalization than those with Delta, and 70% less likely to have severe disease. But other findings in those data seem to contradict the government presentation I featured in my last post, which said hospitalized Omicron patients were half as likely as Delta patients to need intensive care or die. In this new manuscript, which looks at both variants simultaneously instead of comparing today’s Omicron with summertime Delta, there was only a non-significant 30% difference in severe disease. The outcome measures are different, so the discrepancy is hard to interpret.
We’ve been made to believe that Omicron is just a bad cold, largely because of those low hospitalization rates. Unfortunately the story is different if you look outside South Africa, a country where the population is young and 70% have already been infected with SARS-CoV-2. As compared with Delta, the hospitalization rate with Omicron is only two-thirds lower in Scotland and Denmark, and a mere 40-45% in England.
Some more discouraging Omicron news that contradicts previous reassurances: a colleague who performs rapid tests here in Rome told me they’ve had to change their reagents, because the ones that were good for detecting Delta weren’t picking up the new variant. An FDA study has now confirmed her observations for professionally-taken swabs, and home tests likely perform even worse. Antigen tests were already lousy enough at picking up previous variants! Alas, now we have hints from both South Africa and the USA that even PCR tests might be less accurate with Omicron.
Words, wise and otherwise
 |
| Dr. David Kessler |
Two Washington Post journalists have written a very sensible article on what to do if you test positive. Not much new for my readers, but it’s nice to have the advice set out so clearly.
Unfortunately one authoritative source has in my opinion fallen down on the job. Dr. David Kessler, former FDA head and Biden’s chief science officer for the COVID response, said on the Rachel Maddow show (just before minute 3) that people with mild symptoms suggestive of COVID-19 should wear a mask but not bother to get tested. I find this advice irresponsible. Masks are less than perfect, and Omicron is extraordinarily contagious. No mask can fully protect the patient’s family members and workplace contacts, some of whom may be at high risk for severe disease. Any person who has symptoms must get a PCR test, and pending the results must not only wear a mask but stay home in complete isolation until the result comes back negative; if the result is positive, they should keep it up for 10 days.
A tale of three countries
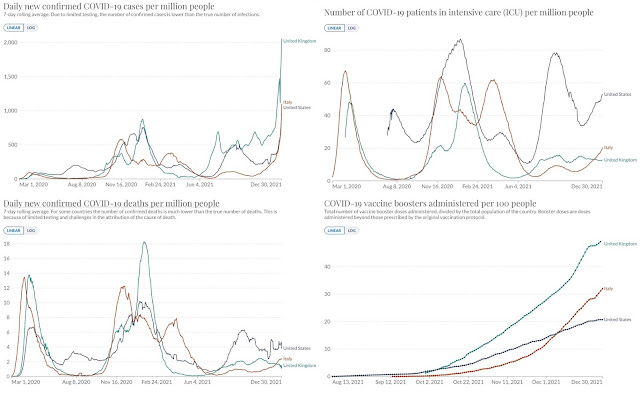
Italian, UK, and US figures (from Our World In Data) provide an object lesson in the value of vaccination. The number of new infections cases per capita is now nearly identical in the US and Italy, and twice as high in the UK. But Italy has about a third as many COVID-19 patients in ICUs as the US, and half as many deaths – and rates in the UK are much lower yet. That’s the vaccines doing their job. The US has more bad outcomes because so few residents are “fully vaccinated” (a near-obsolete term): 61%, far below Italy’s 78% and the UK’s 69%. And the UK has kept those bad outcomes down still further by boosting like mad: nearly half of the entire population has already received a booster shot.
Like everybody, Italy is struggling with case surges by instituting one set of new policies after another. The main change in its latest set, outdoor masking everywhere, is just silly. But some measures are sensible: reintroducing some capacity limits, making KN95 masks (=N95=Ffp2) obligatory for entertainment venues and city transport, cutting the duration of vaccine Green Passes from 9 months to 6, urging boosters after just 4 months, and closing discotheques until February (though cheating is expected).
Probably the best recent Italian innovation is the “Super Green Pass” reserved for people who are vaccinated or post-COVID-19, now required for restaurants (even outside), ski lifts, cinemas, gyms, outdoor sports, weddings and other ceremonies, conferences, museums, hotels, even coffee bars and city buses. But the only thing the Super Green Pass can possibly do is to encourage vaccination. It’s working, but vaccination can’t possibly impact case numbers until masses of repentant novaxers are weeks after their second shot, which won’t be true for months, and without a booster shot vaccines do little to stop Omicron. What Italy needs now is tougher measures: curfews, closing restaurants in the evening, remote work, and limits on private gatherings. And if infections keep raging, lockdown.
The latest COVID-19 papers to hit the dust
- An article claiming to prove that lockdowns don’t work
- A conference poster claiming vaccines damage the heart
- An article claiming vaccination caused miscarriages
- A trumped-up metaanalysis of treatment with granulocyte-macrophage colony-stimulating factor antibodies
- Two Egyptian studies of favirapir treatment (a hydroxychloroquine study from the same group was pulled earlier)
- Yet another ivermectin article
- A paper from notorious anti-vaxers vastly exaggerating the risk of myocarditis after mRNA vaccines (the real data among adolescents are already worrisome enough)